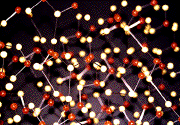
The idea that three-dimensional networks can be seen as two-dimensional nets is most easily seen by looking at pictures. Here is a standard three-dimensional "ball-and-stick" model of a net:

That model implicitly assumes the net is embedded in three-dimensional euclidean space (for example, all balls are connected by straight edges).
Here is a model of a natural zeolite, analcime:

This model contains4-rings, 6-rings and 8-rings.
Exactly the same arrangement of rings and connections can be realised by the simple tiling of the periodic gyroid surface by 4-rings and 6-rings (marked by the black lines):

In other words, if the underlying gyroid surface were "dissolved", leaving only the net marked by black lines, that would be the same net as that shown in the previous picture. So, the periodic network can be viewed as a three-dimensional net, OR a curved (hyperbolic) two-dimensional net.
Another example. The zeolite, known as faujasite. The picture shows the net, and the two-dimensional "embedding" surface which the net can be projected onto, the D-surface:

For more examples, see the page maintained by Stuart Ramsden...
A final example is an interesting example illustrating the sociology of science "discovery". Two structures are shown below, from 1946, published by Holohan, Gibson and Riley (L. Chem. Soc., 1946, p. 456) as possible structures for novel framework forms of graphite (carbon). Almost 50 years later, the idea of novel "hyperbolic" graphites (christened "schwarzites", as opposed to the elliptic fullerenes) was mooted (A.L. Mackay and H. Terrones, Nature, vol. 352, p. 762 (1991)).

It turns out that among of the most stable schwarzite forms are the original framework graphites (now called "polybenzene", M. O'Keeffe et al., Phys. Rev. Lett., vol. 68, p. 2325 (1992))! Schwarzites, then, are not a new construction. Rather, the language describing these structures is new - two-dimensional and hyperbolic, rather than three-dimensional (and flat, or euclidean).
The same dichotomy of dimension and curvature can be found in notions of polyhedra. AF. Wells and H.S.M. Coxeter introduced the notion of "infinite" (or "skew") polyhedra. These are most simply seen as tilings of sponges - such as triply periodic minimal surfaces - with flat polygonal faces.
They are then simple extensions of conventional (convex) polyhedra, which are tilings of the sphere (or a topologically equivalent surface). A good example is the polyhedral packing of icosahedra and octahedra in the alloy structure, WAl12.

This can be seen as a simple triangulation of the D-surface, with 7 triangles sharing each vertex! This description follows at once if seven triangles are placed around each vertex (two such vertices are surrounded by green arrowed loops).

The resulting sheet resembles a crinkled sheet, and as it grows it naturally forms proto-tunnels (yellow arrows). If these tunnels are arranged in the tetrahedral array of the D-surface, the resulting network is that of the edges and vertices of the polyhedral packing characteristic of WAl12.
Alternatively, the triangular faces of the WAl12 polyhedral packing fold onto the D-surface, if all shared faces common to an icosahedron and neighbouring octahedron are removed (forming channels).
So, here too, the notion of dimensionality and curvature is not an absolute one, and the usual three-dimensional euclidean polyhedral packing can be interpreted instead as a close-packing of vertices (giving triangular faces) on a hyperbolic two-dimensional sheet.
A number of other polyhedral packings can be understood similarly. For example, the "pyrochlore" structure can be constructed from face-sharing octahedra alone. If shared faces are removed, the resulting infinite polyhedron contains eight triangles about each vertex (instead of seven in the previous example - pyrochlore is thus topologically more dense than WAl12). In this case also, the triangulation lies in the D-surface. A "classical" three-dimensional view of the vertex arrangement in the pyrochlore structure is shown below. In this model face-sharing octahedra are not evident - a half the octahedra are missing - although all the pyrochlore vertices are contained in the vertex-sharing octahedral packing shown).

The structures describe dhere are all reasonably low-density forms, as they indeed must be if they tile intersection-free triply periodic hyperbolic surfaces. The challenge now is to see whether that two-dimensional description can be usefully extended to denser structures, which (presumably) tile intersecting hyperbolic surfaces.
Posted Nov. 1997.